Services for Protein Biophysics
Protein-protein and protein-ligand interactions are the backbone of almost every vital cellular process and drug discovery project.
We have developed a portfolio of tailor-made services that allow for the precise, robust, fast, and cost-efficient analysis of almost any protein interaction: Proteins and peptides, DNA, RNA, aptamers, small molecules, molecular fragments, ions, viruses, VLPs, and artifical particles. The state-of-the-art biophysical methods MST, nanoDSF, BLI, and ITC are available for in-depth comprehensive interaction analysis, as well as fast and cost-efficient high-throuhput screening.
Moreover, we are able to analyze protein stability, thermal and chemical protein unfolding, protein denaturation and aggregation, as well as buffer optimization, using the state-of-the-art, label-free, in-solution nanoDSF method.
If you are interested in specific example applications, please have a look at our application notes. You can also use the contact form below to ask us anything about the services outlined below. We will get back to you shortly with a solution made specifically for your request!
Services for Protein Interactions
With our tailor-made biophysical assays, interactions between a protein and almost all kinds of relevant ligands can be analyzed: Proteins, peptides, DNA, RNA, aptamers, small molecules, molecular fragments, ions, lipids, virus-like-particles, and artifical particles.
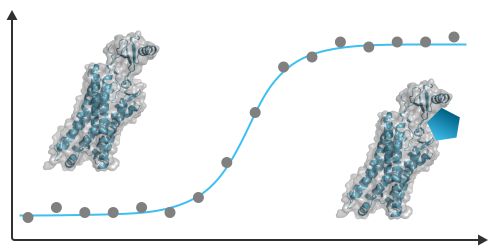
Steady-state Binding Assay - MST
Determine the steady-state affinity (KD) of your protein or peptide to its natural ligand or to other possible targets, including ions, small molecules, fragments, peptides, DNA, RNA, proteins, or synthetic particles.

High-throughput screening - MST, nanoDSF
Analyze up to 10 000s of small molecule compounds or molecular fragments for binding to a protein target. Fast, precise, cost-efficient, and with the lowest sample consumption on the market.
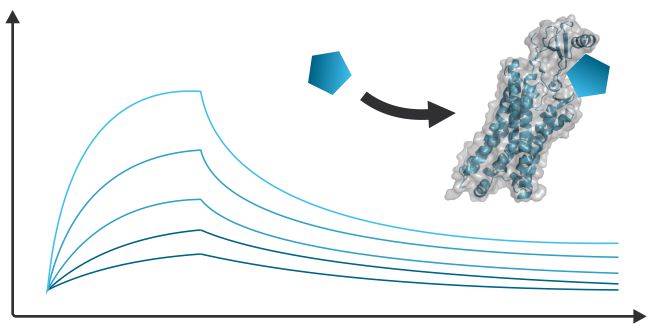
Dynamic Kinetic Assay - BLI
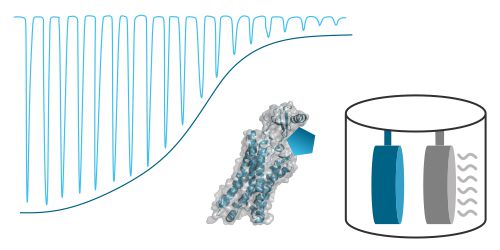
Thermodynamic Profiling - ITC
Assays in Biological Liquids - MST
Competition Assay - MST, BLI
Services for Protein Stability and Unfolding
Information about thermal protein stability is essential in many cases. For example, high protein stability is an important factor for mutational scanning of a protein. Also, therapeutic and diagnostic applications often require proteins to be very stable under the most diverse conditions.
Our precise, versatile, fast, and cost-efficient nanoDSF platform allows for the determination of protein stability in a wide range of applications outlined below. Moreover, nanoDSF allows for the label-free, in-solution analysis of thermal and chemical protein unfolding as well as the detection of protein denaturation and protein aggregation. Importantly, no extrinsic fluorescent dye like in traditional DSF methods is required.

Protein Stability Buffer Screening
Find the optimal buffer or buffer additives for your protein in a pre-formulation phase buffer screening. We can work with any custom-made buffer you require. Alternatively, the 2bind FORMOscreen can be used, offering 96 individual buffers derived from antibody formulations.
Thermal Shift Binding Assay
Test binding of small molecules, RNA, DNA, peptides, or fragments to proteins by measuring shifts in their thermal stability.

Long-term Storage Optimization
Protein Batch Comparison