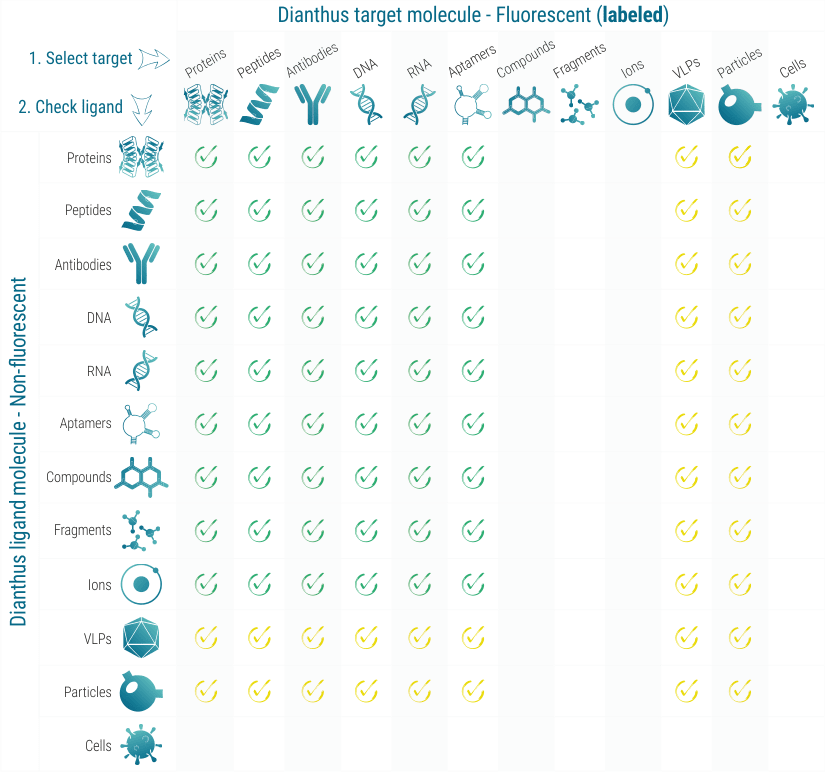
Molecular interactions that can be measured with Dianthus
The Dianthus instrument relies on the tried-and-tested measurement principle TRIC (temperature-related intensity change of fluorophores), which has been proven in MicroScale Thermophoresis (MST) for over a decade. Thus it works with almost all classes of target molecules. In comparison to classical MST, however, a fluorescent label on the target molecule is mandatory, because no label-free measurement is possible. Large target or ligand molecules (e.g. virus-like or other particles) can be challenging, because they have to be kept in solution in the 384-well plates the instrument uses and must not sink down to the well bottom. As in classical MST, the fluorescent target molecule is kept at a constant concentration and the non-fluorescent ligand molecule is titrated across a large concentration range.
If you are uncertain whether your type of interaction can be measured with Dianthus or if you want to use the 2bind Dianthus service just leave us a message:
What Dianthus can tell
Single-dose screen
Dianthus will tell you whether a target and ligand generally interact from a single-dose experiment.
Steady-State Affinity
Dianthus will tell you the precise and true steady-state affinity (dissociation constant KD) of your interaction.
Protein Aggregation
Dianthus will tell you whether your protein samples aggregate or if aggregation is induced by ligand binding.
Ligand competition
Dianthus will tell you competitive effects between multiple binders of your targets.
- Drug discovery (compound-based and fragment-based)
- Drug discovery (protein-targeted and RNA-targeted)
- Protein characterization
- Additive screening
- Enzyme-inhibitor screening
- Drug discovery (compound-based and fragment-based)
- Drug discovery (protein-targeted and RNA-targeted)
- Protein characterization
- Additive screening
- Enzyme-inhibitor screening
- Protein and enzyme quality control
- Buffer screening and optimization
- Protein storage optimization
- Ligand-induced aggregation testing
- Complex binding analysis
- Mode-of-action analysis
- Competitor screening
- PPI-disruption analysis
Advantages of Dianthus
2bind Dianthus Services
2bind offers a variety of services using the Dianthus instrument. Benefit from fast and precise analysis of molecular interactions.
Enjoy minimal sample consumption (just nM concentrations and µL volumes) and robust analysis methods. The 2bind Dianthus services can be combined as you choose for all questions in drug discovery, antibody development, protein biophysics and analysis, as well as aptamer characterization.
Dianthus Technology and FAQs
Overview
The Dianthus technology (MST Service) is a biophysical technique that measures the strength of interaction (affinity) between two molecules by detecting variations in fluorescence signals as a result of an IR-laser induced temperature change. The range of the variation in the fluorescence signal correlates with the binding of a ligand to the fluorescent target. By that it allows for quantitative analysis of molecular interactions in solution on the microliter scale with high sensitivity.
The Dianthus technology relies on the so-called TRIC effect. TRIC stands for Temperature-Related Intensity-Change and describes how the fluorescence intensity of a fluorophore depends on the local temperature of the solution. For the vast majority of fluorophores, the fluorescence intensity decreases with increasing temperature. Most importantly, however, the extent of the temperature dependence is strongly related to the chemical environment of the fluorophore.
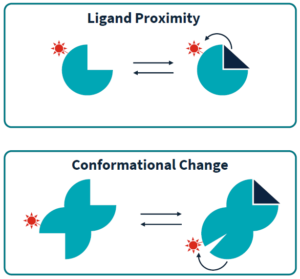
In order to measure such TRIC effect changes, a target molecule is labeled with a special fluorescent dye and a precise and brief heat-gradient is induced via an infrared laser. The TRIC signal is affected by the fluorophores electronic microenvironment, which changes upon (i) binding of a ligand to the target molecule and (ii) conformational changes of the target molecule upon binding of a ligand.
Technology
The Dianthus technology is based on the TRIC effect (temperature-related intensity-change of fluorescent molecules). These fluorescence changes are very sensitively dependend on the electronic microenvironment of the fluorescent molecule. Thus, if a special fluorescent dye is coupled to a target molecule, its TRIC effect will react to changes by ligand binding or by ligand bindig-induced conformational changes.
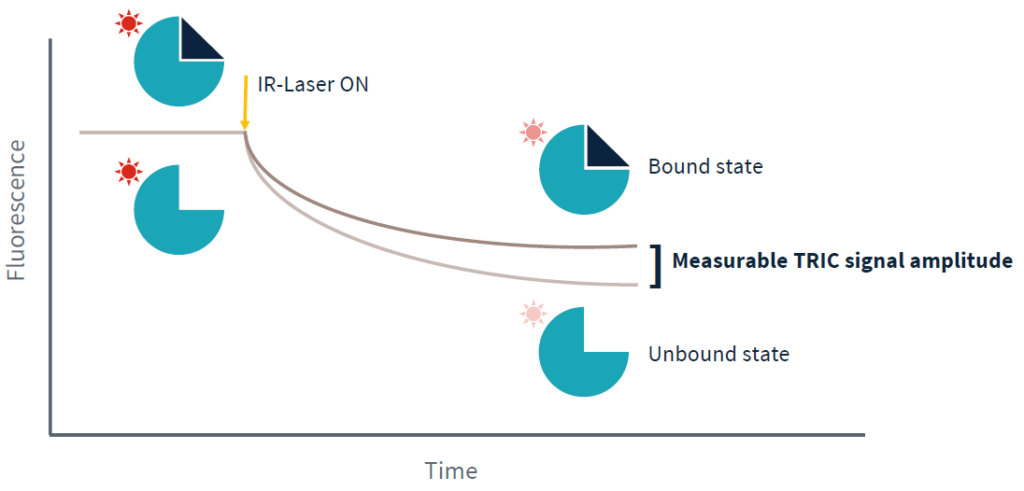
The figure above gives an overview of a Dianthus experiment. In contrast to classical MST, where the samples are present inside small glass capillaries, the Dianthus technology uses 384-well plastic plates with transparent bottoms. In 12 adjacent wells, mixes of the fluorescently-labeled target molecule and increasing concentrations of the ligand molecule are placed. As in classical MST, an infrared laser is used to generate a precise temperature gradient while an LED is used for the excitation of fluorescent molecules in the wells. The laser-induced temperature gradient leads to the TRIC-based changes of the fluorescence of the labeled target molecule, which is recorded by fluorescence optics. Different TRIC-responses are obtained for different states of the target molecule, i.e. ligand-unbound and ligand-bound states.
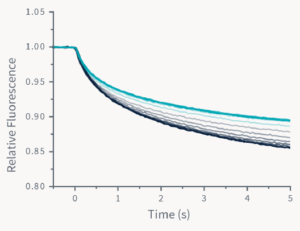
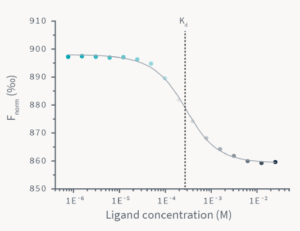
The comparison of the TRIC traces for all 12 ligand-concentration points reveals that there is a gradual change in the TRIC response that correlates with the ligand concentration. The TRIC traces can finally be transformed to a classical dose-response curve, which can be fitted to various models to yield the steady-state affinity of the analyzed molecular interaction.
Typical applications
Typical applications of Dianthus include:
- Hight-throughput target-ligand interaction screenings
- Drug discovery (protein-targeted and RNA-targeted)
- Drug discovery (compound-based and fragment-based)
- Steady-state binding affinity assay
- Steady-state binding affinity assay in biological liquids
- Sandwich assays (1 target, 2 ligands)
- Competition assays
- Binding affinity assays with multiple binding partners
For more information, please visit our Application Database. The different service areas can be found under MST Services.
Compatible Fluorescent Dyes
The Dianthus technology required labeling of the target molecules with a fluorescent dye in the 650 nm wavelength excitation range. Depending on which MST Service you require, different fluorescent methods and/or labelings are possible. Most commonly, the labels listed in the table below are used. 2bind performs the labeling of your target protein for you with all possible dyes. Another option is to fuse a potential target protein with an intrinsically fluorescent protein fluorophore such as RFP or the like.
In the case of DNA or RNA target molecules direct covalent linkage of fluorophores (e.g. Cy5) has proven well.
The following table gives an overview over the most commonly used fluorophores in Dianthus. Please keep in mind that, in principle, every fluorophore can be used as long as its excitation and emission wavelength ranges match the ones of the fluorophores listed here.
| Fluorophore | Excitation (nm) | Emission (nm) |
|---|---|---|
| Cy5 | 649 | 670 |
| NT-647 (RED) | 650 | 680 |
| Alexa647 | 652 | 668 |
| NT-RED 2nd generation | 650 | 670 |
Advantages
The Dianthus technology offers a number of great advantages over other biophysical methods for determining the affinity of a molecular interaction. Follow this link for an overview over our possible services.
- Low sample consumption → minimum of only 6 µl is required per sample
- Free choice of assay buffers → also biological liquids possible such as serum or cell lysate
- Very short analysis time → short analysis time enables high throughput
- Real-time quality controls → online aggregation, precipitation, and sticking controls
- Wide temperature range → analysis possible from 20°C to 45°C
- No immobilization required → measurement is done truly in solution
- Wide concentration range → affinities can be analyzed in the pM-mM range
- Wide molecule size range → from 100 Da to 1 MDa
For a comparison of Dianthus with other biophysical techniques, please refer to our Technology Comparison Guide.
FAQ – General
What kinds of molecular interactions can I measure?
You can measure bi-molecular interactions between any kind of molecule: protein, RNA, DNA, small molecule compounds, lipids, carbohydrates (See figure on top of this page).
Is it possible to analyze interactions with aptamers?
Yes. Analyzing molecular interactions involving aptamers or other DNA- or RNA-molecules is no problem. Usually, Dianthus measurements with aptamers are straight-forward, because these DNA molecules can be synthesized directly with fluorescent labels at their 5′ or 3′ end. Most commonly, Cy5 or Alexa647 are used as aptamer or DNA fluorophores.
More information on this topic is available in our Aptamer Services.
What information do I get from a Dianthus MST measurement?
Dianthus is not only able to determine the affinity and binding strength of a molecular interaction, but also allows for assessing other physical parameters such as aggregation, precipitation, enthalpy (van’t Hoff plot), slow enzyme kinetics, and oligomerization.
For specialized kinetics measurements, consider Biolayer Interferometry. If you are interested in the thermodynamics of a molecular interaction, Isothermal Titration Calorimetry is an option. For an in-depth analysis of protein aggregation, protein stability and protein unfolding, take a look at the nanoDSF technique.
What type of fluorescent dyes can I use?
The Dianthus instrument can detect typical “red” fluorescent dyes. Please see above (“Compatible Fluorescent Dyes”) for more information on the specific dyes that can be used.
Is it possible to measure without labelling a molecule?
No, in contrast to classical MST, the Dianthus instrument is not available in a “label-free” format and cannot detect intrinsic tryptophan fluorescence.
Can I measure binding kinetics with Dianthus?
Dianthus measures equilibrium binding constants (steady-state affinity). Typically, samples are inserted in the instrument after the binding reaction reaches chemical equilibrium. Measurement of kinetics is possible if the equilibrium is not reached quickly. On average, the kinetics of reactions that take more than 20 minutes can be evaluated.
An in-depth kinetics analysis of molecular interactions is possible with Biolayer Interferometry.
Can I measure thermodynamics with Dianthus?
Dianthus measures equilibrium binding constants (steady-state affinity) at a given temperature. By performing such experiments at different temperatures, the binding enthalpy can be derived from van’t Hoff analysis.
An in-depth thermodynamics analysis of a molecular interaction is possible with Isothermal Titration Calorimetry.
FAQ – Samples
Can I check for the quality of my samples?
Yes! Direct intrinsic quality controls available in the Dianthus setup allow for detecting protein aggregation as well as adhesion and sticking effects of the assay samples to the 384-well assay plates.
An in-depth analysis of the stability and aggregation behavior of your protein samples can be done with nanoDSF.
Is it possible to measure the binding of small molecules?
Yes, we are able to measure molecular interactions involving small molecules with the same high sensitivity and quality as for protein-protein, protein-DNA or protein-vesicle interactions. For more information on Dianthus and small molecule interaction analysis please have a look at our Drug Discovery Services.
What are the required concentrations for small molecules?
Dependent on the fluorophore (fluorescent label or intrinsic tryptophan fluorescence), between 1 – 100 nM of the labeled target molecule can be used. This target is titrated with the unlabeled small molecule compound in the range of ± factor 10 of the expected affinity (dissociation constant). For standard applications, 20 µl of sample material are used per data-point.
My protein is only stable at high ionic strength. Can I still test for binding with Dianthus?
If you are not sure whether your sample protein really requires high ionic strength buffers, it is possible to investigate protein stability, thermal unfolding, and its aggregation behavior using nanoDSF. We offer a variety of Services in this respect.
I want to measure binding to a Mega-Dalton (MDa) molecule. Is this possible?
Given that the large target molecule does not sediment or sink down to the bottom of the 394-well assay plates during the experiment, the analysis of large molecules is possible with the same considerations as for classical MST.
FAQ – Assay Conditions
Are there any limitations to the buffers or additives I can use?
No. You can use practically any buffer and any additive as long as it does not negatively affect the stability or homogeneity of your sample. Additives that affect the viscosity of the sample solution should be kept at a constant concentration. As the measurements take place in 384-well plastic plates (and not in glass capillaries as in classical MST), organic solvents cannot be used.
A great way to check the tolerance of your protein samples for a vast number of additives is nanoDSF, a method that analyzes the thermal stability and unfolding of proteins. This technique can be used for buffer and additive screenings.
Can I measure in biological liquids like serum or lysates?
If you are interested in the stabilty of your protein sample, this can be determined using our nanoDSF technology.