Biophysical Characterization of Protein-Aptamer Interactions using MicroScale Thermophoresis (MST)
Clemens Entzian, Thomas Schubert
2bind GmbH, Regensburg, Germany
MST Technology
MicroScale Thermophoresis (MST) is an optical fluorescent method. It records the changes in fluorescence of a target molecule as a function of temperature and the concentration of the cognate ligand molecule. Thus, in MST, the target must be fluorescent and the ligand must not be fluorescent in the same wavelength range as the target. The fluorescence changes are the result of two effects:
First, molecules exhibit directed movement along temperature gradients in solution, an effect called thermophoresis (Duhr & Braun, 2006). Such thermophoretic movement of a molecule is characterized by its size (hydrodynamic radius), its surface charge, and its hydration shell. All three parameters can be affected by binding of a ligand molecule. Thus, a concentration-dependent titration of the target molecule’s ligand induces changes in the thermophoretic movement. These changes translate in quantifiable, spatial fluorescence changes, which are easily tracked optically via the target fluorescence.
Second, fluorescence is a function of the temperature. Thus, the fluorescence of the target molecule varies with the temperature (Baaske et al., 2010). This temperature-dependence is additionally affected by the local molecular surroundings of the fluorophore. Thus, binding of a ligand molecule to the fluorescent target can change the chemical environment of the target fluorophore and thus the overall detected temperature-dependence of the fluorescence. This effect is known as TRIC (temperature-related intensity change; Gupta, Duhr & Baaske, 2018).
The thermophoresis and TRIC signals are additive and thus both contribute to the high sensitivity and robustness of MST measurements towards molecular binding events of all kinds. Thus, MST can be used for determining the affinity and binding strength of almost any kind of molecular interaction with very low sample consumption and very high sensitivity.
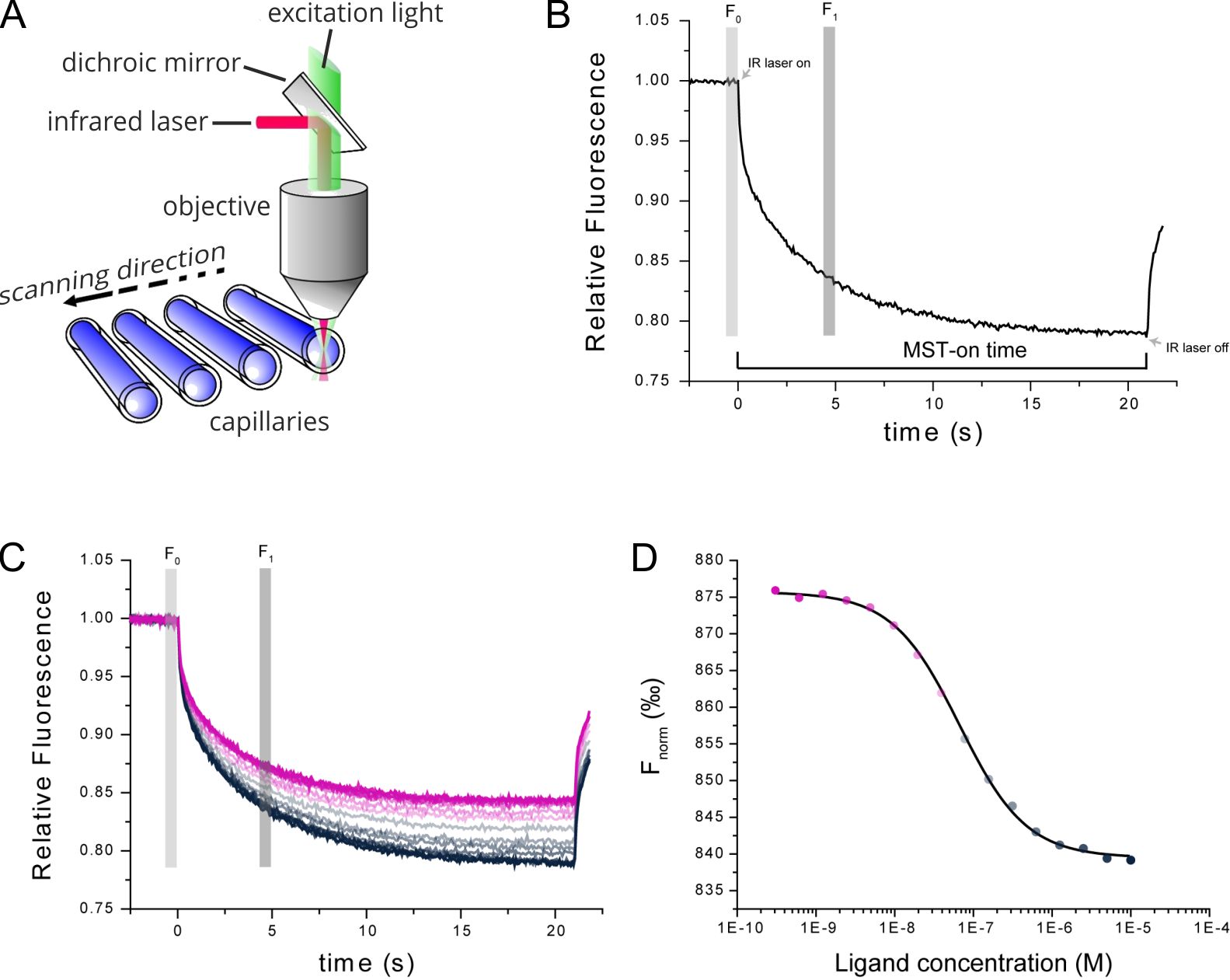
Figure 1. Technical MST setup. (A) MST measurements take place in small glass capillaries. Infrared and fluorescence lasers are used for generation of the MST effect and sample tracking. (B) TRIC and thermophoresis together account for a time-dependent change in fluorescence upon infrared-heating of the sample capillaries. (C) Multiple MST traces are recorded for different mixture ratios of target and ligand molecules. (D) Dose-response analysis of the MST traces allows for determination of the steady-state affinity of the target-ligand interaction.
Introduction: Aptamer interactions
Monoclonal antibodies (mAbs) are the gold-standard of macromolecule-based drug treatments of many diseases. However, mAb development is a time and cost-intense process. Moreover, the therapeutic use of mAbs is somewhat limited due to mAb stability, specifity, and deliverability. Nucleic acid aptamers are a promising alternative to antibodies because of smaller size, reduced development cost, up to equal sensitivity, and reusability. Aptamers are short DNA or RNA sequences binding their target via a unique tertiary structure. The assessment of binding characteristics of aptamers towards their targets is the pivotal initial step in the selection of aptamers (Entzian & Schubert, 2016).
Here, we demonstrate the potential of MST for examining the binding affinity of RNA and DNA aptamers. We show different assay format and types that allow for a fast and precise aptamer binding characterization. Aptamers can be easily used in MST as the fluorescent target, because they can be readily synthesized with appropriate fluorescent dyes attached (e.g. Cy5, Alexa-647).
RNA-Aptamers
Example: Extracellular signal-regulated kinase 2 (ERK2) vs C5.71 Aptamer
ERK2 is a major determinant in the control of cell growth, cell differentiation, and cell survival. The ERK pathway is often upregulated in human tumors and, as such, ERK2 represents an attractive target for anticancer therapy (Kohno & Pouyssegur. 2006). Here, we analyzed the binding affinity of the C5.71 aptamer against ERK2 using an MST assay. Although aptamers can be easily labeled with a fluorescent dye during synthesis, it is also possible to use an aptamer in the completely native form.
In this case, the aptamer target (here the ERK2) has to be labeled with a fluorescent dye. The C5.71 aptamer showed a moderate binding affinity to ERK2 with a KD-value in the low micromolar concentration range (KD: 171.1 ± 36.0 µM) (Figure 2).
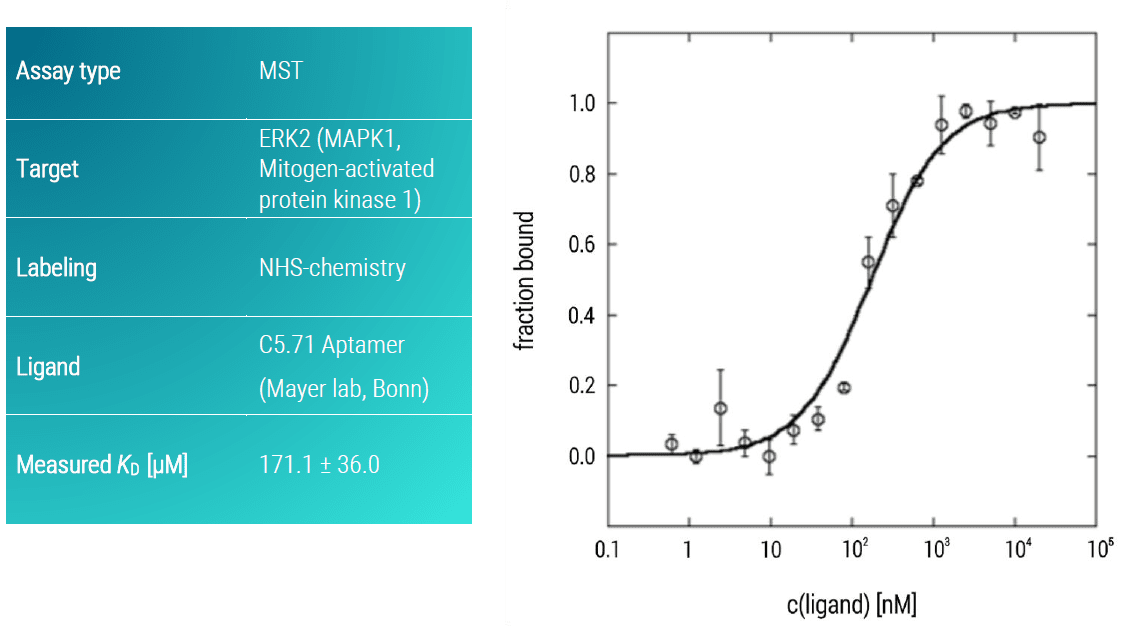
Figure 2: MST dose-response curve of ERK2 and C5.71 aptamer. Data represent the mean (+/- standard deviation) from three independent experiments and were fitted to a KD‑binding model assuming a 1:1 binding stoichiometry.
DNA-Aptamers
Example: β-CBA II aptamer vs β-Conglutin
Conglutins represent the major storage protein family in Lupin seeds and are part of carbon, sulphur, and nitrogen supply for energy metabolism of the germinating seedling (Foley et al. 2015). Thus, β-conglutin has been reported to be one of the most frequent causes of severe food-associated anaphylaxis (Rubio et al. 2016). Here, we determined the binding affinity of the β-CBA II aptamer against β-conglutin using an MST assay. In this case, the aptamer was synthesized with a Cy5 fluorescent dye on its 5’ end. The aptamer showed a strong binding affinity towards β-conglutin with a KD-value in the low nanomolar concentration range (KD: 11.8 ± 2.4 µM) (Figure 3).
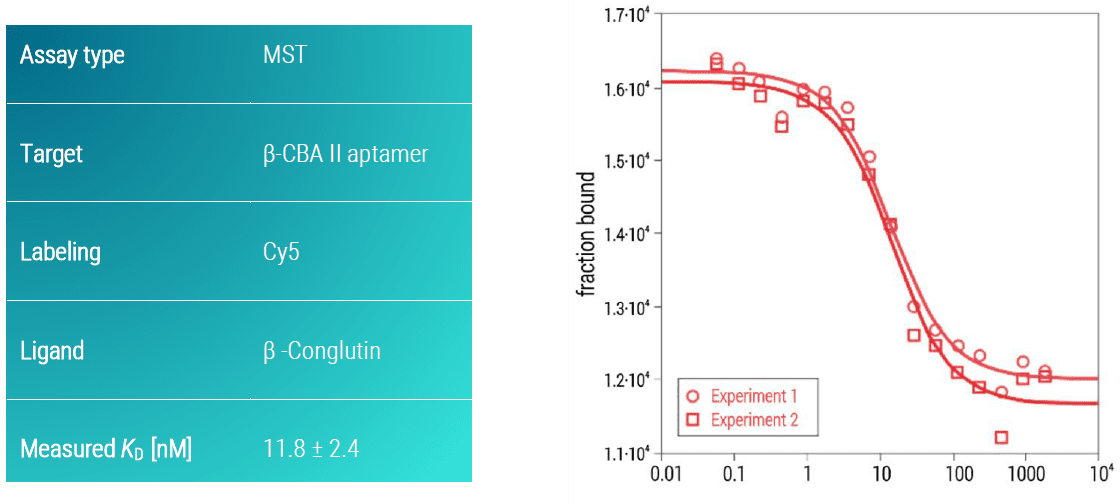
Figure 3: MST dose response curve of β-conglutin and β-CBA II aptamer. Data represent the mean (+/- standard deviation) from two independent experiments (shown individually in the graph) and were fitted to a KD‑binding model assuming a 1:1 binding stoichiometry.
Example: HD1 & HD22 aptamers vs Thrombin
The serine protease thrombin plays a crucial role in coagulation, inflammation, and cellular proliferation events (Siller Matula et al., 2011). Due to its regulatory function in the conversion of fibrinogen to fibrin, and concomitant platelet aggregation, thrombin is an important therapeutic target (Breitspeicher et al., 2016). Here, we analyzed the binding affinity of two DNA aptamers (HD1 & HD22) for thrombin using an MST assay (Figures 4, 5, blue curves). The aptamers were synthesized with a Cy5 fluorescent dye attached to their 5’ ends. Both aptamers showed strong binding affinities to thrombin under aequeous buffer conditions (HD1, KD: 104.7 nM; HD22, KD: 0.71 nM).
Importantly, the developed MST assay also allowed for assessing the specificity of the two aptamers for thrombin in a close-to-native setting. For this, instead of buffer, a 50% fetal calf serum solution was used (FCS, Figures 4, 5, red curves). Whereas the HD22 aptamer was not influenced by the presence of serum (almost unchanged KD value of 132.7 nM), the HD1 aptamer showed a drastic decrease in its affinity of approximately 100-fold (KD value in presence of FCS 90.5 nM). Thus, this aptamer is significantly affected by off-target effects. In a therapeutic scenario, this aptamer would strongly be impaired in its intended function by binding not only to thrombin but also to other serum proteins. This quick and easy MST assay demonstrated the importance of checking off-target effects of aptamers.
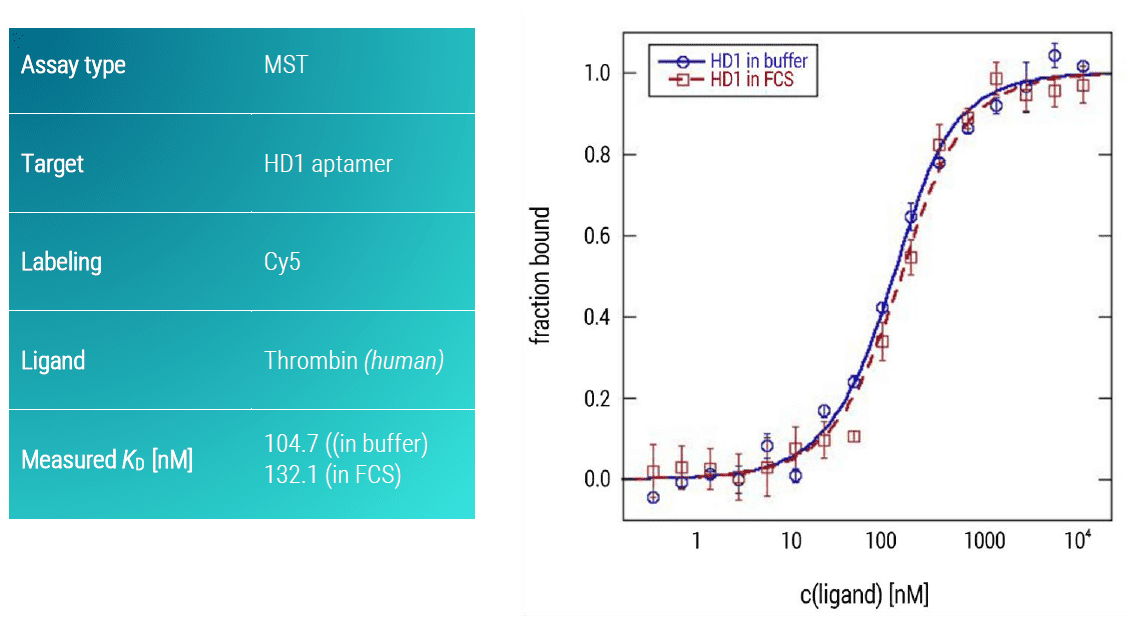
Figure 4: MST dose response curve of thrombin and HD1 aptamer. Data represent the mean (+/- standard deviation) from three independent experiments and were fitted to a KD‑binding model assuming a 1:1 binding stoichiometry.
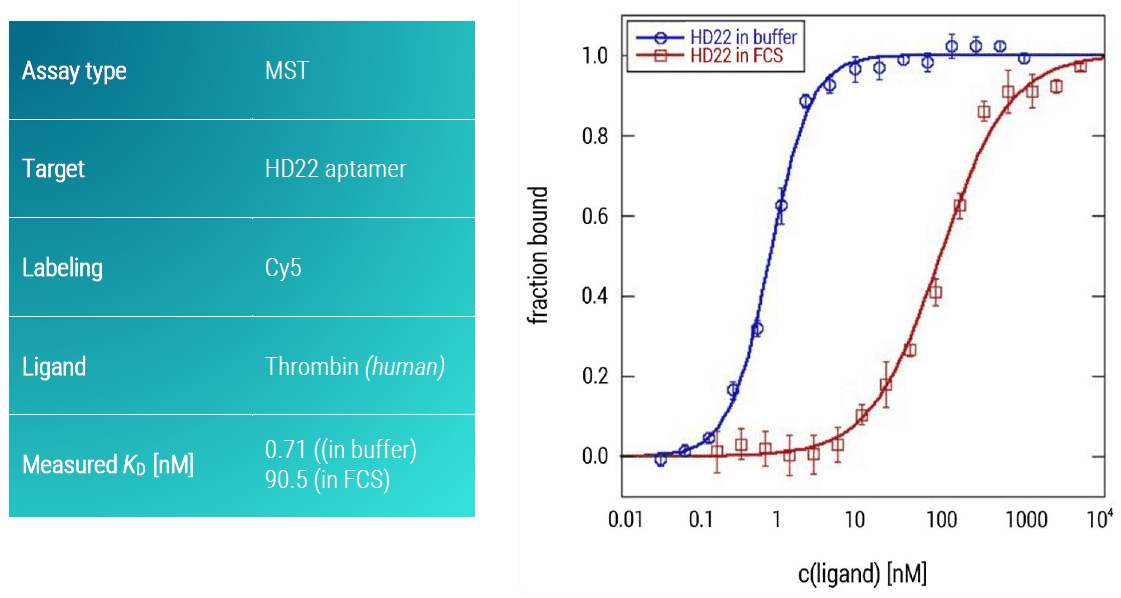
Figure 5: MST dose response curve of thrombin and HD22 aptamer. Data represent the mean (+/- standard deviation) from three independent experiments and were fitted to a KD‑binding model assuming a 1:1 binding stoichiometry.
Conclusion: 2bind MST assays for measuring binding of aptamers to proteins
In this application note we show MST assays that allow for the efficient, rapid, and precise characterization of the binding affinities of RNA or DNA aptamer-protein interactions. With only minor tweaks such assays can be adapted for almost all protein-aptamer interactions.
Additionally, this application note highlights the potential of MST assays for discovering previously unseen off-target effects of aptamers in native-like biological solutions.
Click here for a full list of services for aptamer analysis. Alternatively, see our services for protein analysis. If you like to directly contact us for any kind of service, use the contact form below.
Literature
Baaske et al. Optical thermophoresis for quantifying the buffer dependence of aptamer binding. Angew. Chem. Int. Ed Engl. 2010; 49(15): 2238-2241
Breitspeicher et al. Aptamer Binding Studies Using MicroScale Thermophoresis. Methods Mol Biol. 2016;1380: 99-111.
Duhr & Braun. Why molecules move along a temperature gradient. Porc. Natl. Acad. Sci. USA 2006; 103(52): 19678-19682.
Entzian & Schubert. Studying small molecule–aptamer interactions using MicroScaleThermophoresis (MST) Methods. 2016; 97: 27–34
Foley et al. Analysis of conglutin seed storage proteins across lupin species using transcriptomic, protein and comparative genomic approaches. BMC Plant Biology (2015) 15(106): 1-12.
Gupta, Duhr & Baaske. MicroScale Thermophoresis (MST). Encyclopedia of Biophysics. 2018: 1-5
Siller-Matula et al. Thrombin as a multi-functional enzyme. Focus on in vitro and in vivo effects. Thromb Haemost 2011; 106(6):1020–1033
Kohno & Pouyssegur. Targeting the ERK signaling pathway in cancer therapy. Ann Med. 2006; 38(3): 200-211.
Jauset Rubio et al. β-Conglutin dual aptamers binding distinct aptatopes. Anal Bioanal Chem. 2016; 408: 875–884
