Analysis of Protein-Protein Interactions using MicroScale Thermophoresis (MST)
Clemens Entzian, Thomas Schubert
2bind GmbH, Regensburg, Germany
MST Technology
MicroScale Thermophoresis (MST) is an optical fluorescent method. It records the changes in fluorescence of a target molecule as a function of temperature and the concentration of the cognate ligand molecule. Thus, in MST, the target must be fluorescent and the ligand must not be fluorescent in the same wavelength range as the target. The fluorescence changes are the result of two effects:
First, molecules exhibit directed movement along temperature gradients in solution, an effect called thermophoresis (Duhr & Braun, 2006). Such thermophoretic movement of a molecule is characterized by its size (hydrodynamic radius), its surface charge, and its hydration shell. All three parameters can be affected by binding of a ligand molecule. Thus, a concentration-dependent titration of the target molecule’s ligand induces changes in the thermophoretic movement. These changes translate in quantifiable, spatial fluorescence changes, which are easily tracked optically via the target fluorescence.
Second, fluorescence is a function of the temperature. Thus, the fluorescence of the target molecule varies with the temperature (Baaske et al., 2010). This temperature-dependence is additionally affected by the local molecular surroundings of the fluorophore. Thus, binding of a ligand molecule to the fluorescent target can change the chemical environment of the target fluorophore and thus the overall detected temperature-dependence of the fluorescence. This effect is known as TRIC (temperature-related intensity change; Gupta, Duhr & Baaske, 2018).
The thermophoresis and TRIC signals are additive and thus both contribute to the high sensitivity and robustness of MST measurements towards molecular binding events of all kinds. Thus, MST can be used for determining the affinity and binding strength of almost any kind of molecular interaction with very low sample consumption and very high sensitivity.
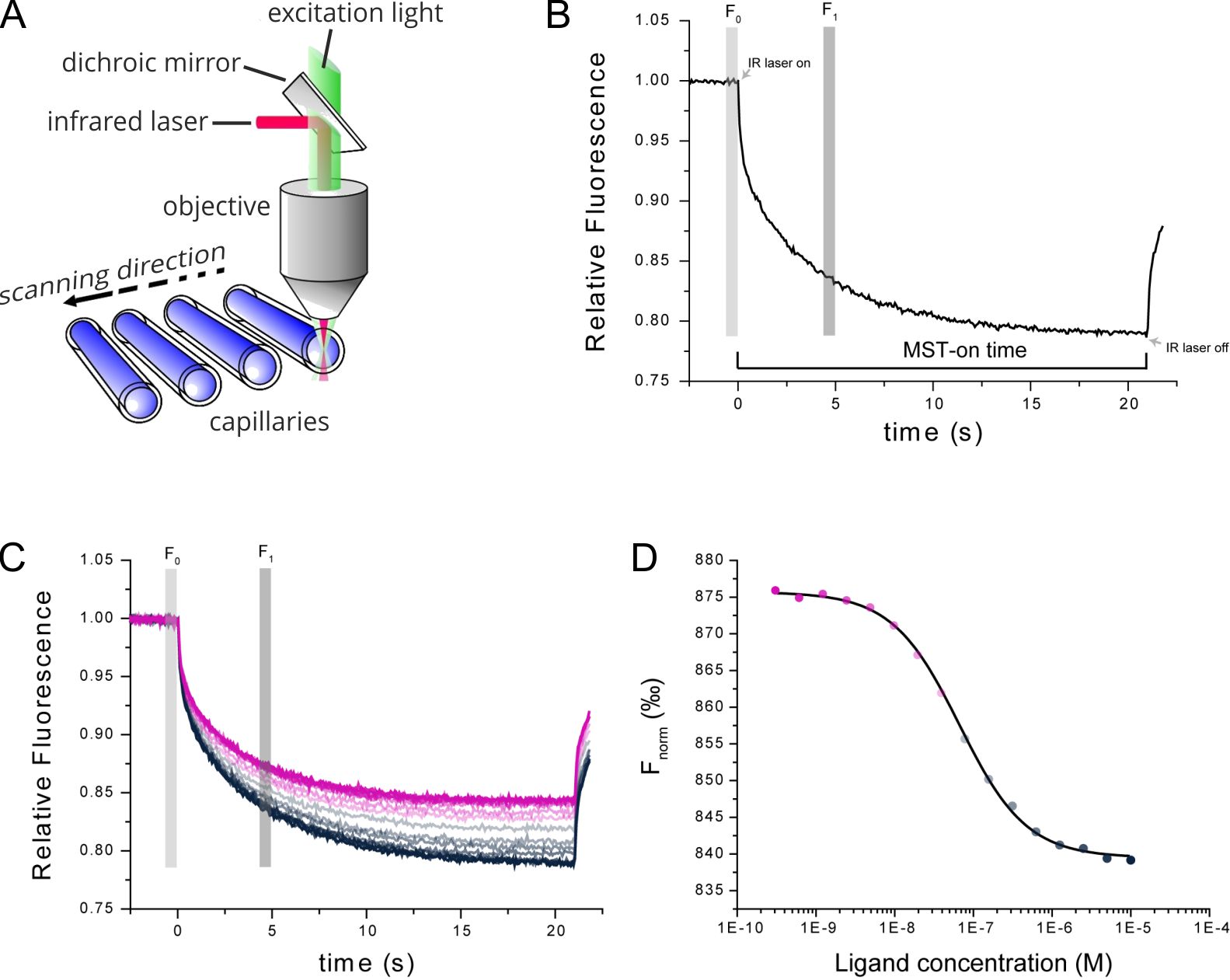
Figure 1. Technical MST setup. (A) MST measurements take place in small glass capillaries. Infrared and fluorescence lasers are used for generation of the MST effect and sample tracking. (B) TRIC and thermophoresis together account for a time-dependent change in fluorescence upon infrared-heating of the sample capillaries. (C) Multiple MST traces are recorded for different mixture ratios of target and ligand molecules. (D) Dose-response analysis of the MST traces allows for determination of the steady-state affinity of the target-ligand interaction.
Introduction: Protein-Protein Interactions
Protein-protein interactions (PPIs) control a variety of biological processes including cell development, cell-to-cell interactions and metabolic pathways. Dysregulated PPIs, especially in signal transduction or epigenetic networks, represent the main reason for disease development and thus deliver a multitude of drug targets. Most analytical methods for the interaction of proteins are either time-consuming (e.g. SPR, ITC), require high amounts of sample (e.g. ITC, SEC), or highly expensive machinery (e.g. mass spectrometry). MST presents an attractive alternative that combines the advantages of speed, ultra-low sample consumption, and high-throughput/cost-efficiency. Here, we used MST to establish experimental setups for fast and precise determination of binding affinity of various PPIs. We focused on three different forms of PPIs: protein-peptide, protein-histone, and protein-nucleosome interactions.
Protein-Peptide Interactions
Example: Extracellular signal-regulated kinase 2 (ERK2) vs ERK2 activation inhibitor peptide I
ERK2 is a major determinant in the control of cell growth, cell differentiation, and cell survival. The ERK pathway is often upregulated in human tumors and, as such, ERK2 represents an attractive target for anticancer therapy (Kohno and Pouyssegur. 2006). Throughout the rational design of bioavailable small molecule drugs, peptides play a pivotal initial role for rapid identification of ligands.
Here, we analyzed the binding affinity of a potential inhibitory active peptide against ERK2 using an MST assay. ERK2 activation inhibitor peptide I presented a strong binding affinity to ERK2 with a KD-value in the low micromolar concentration range (KD: 4.9 ± 1.7 µM) (Figure 2).
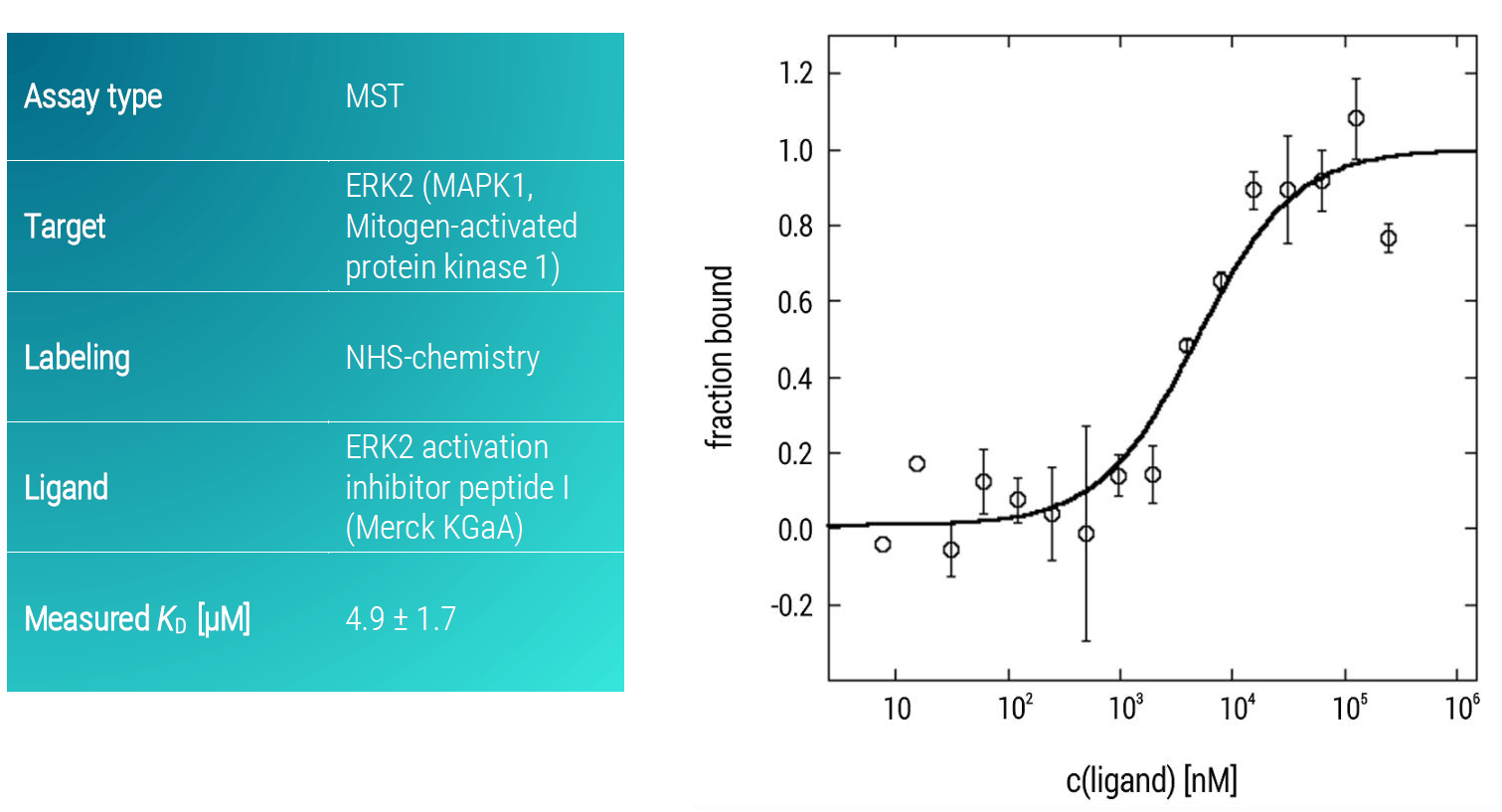
Figure 2. MST dose-response curve of ERK2 and ERK2 activation inhibitor peptide I. Data represent the mean (+/- standard deviation) from three independent experiments and were fitted to a KD‑binding model assuming a 1:1 binding stoichiometry.
Protein-Protein Interactions
Example: Protein-protein interaction between Decondensation factor 31 (Df31) and various core histones
The decondensation factor 31 tethers short, single stranded RNAs to Drosophila chromatin, which there-upon link stably to specific chromatin domains and open the chromatin structure. Therefore, Df31 plays a major role in the opening of Drosophila chromatin and is an important effector in epigenetic networks (Schubert et al. 2012). Here, we analyzed interactions between Df31 and various core histones in an MST assay. For this, Df31 was expressed as a fusion construct to GFP for the required fluorescence tracking.
Df31-EGFP interacted specifically with H3 with a KD of 1.5 μM (±0.4 μM) and bound weakly to H4 with a KD of 12 μM (± 0.6 μM) and did not bind to H2A and H2B (Figure 3). Control titrations of free EGFP against H3 and H4 showed that the GFP itself does not affect the observed interactions.
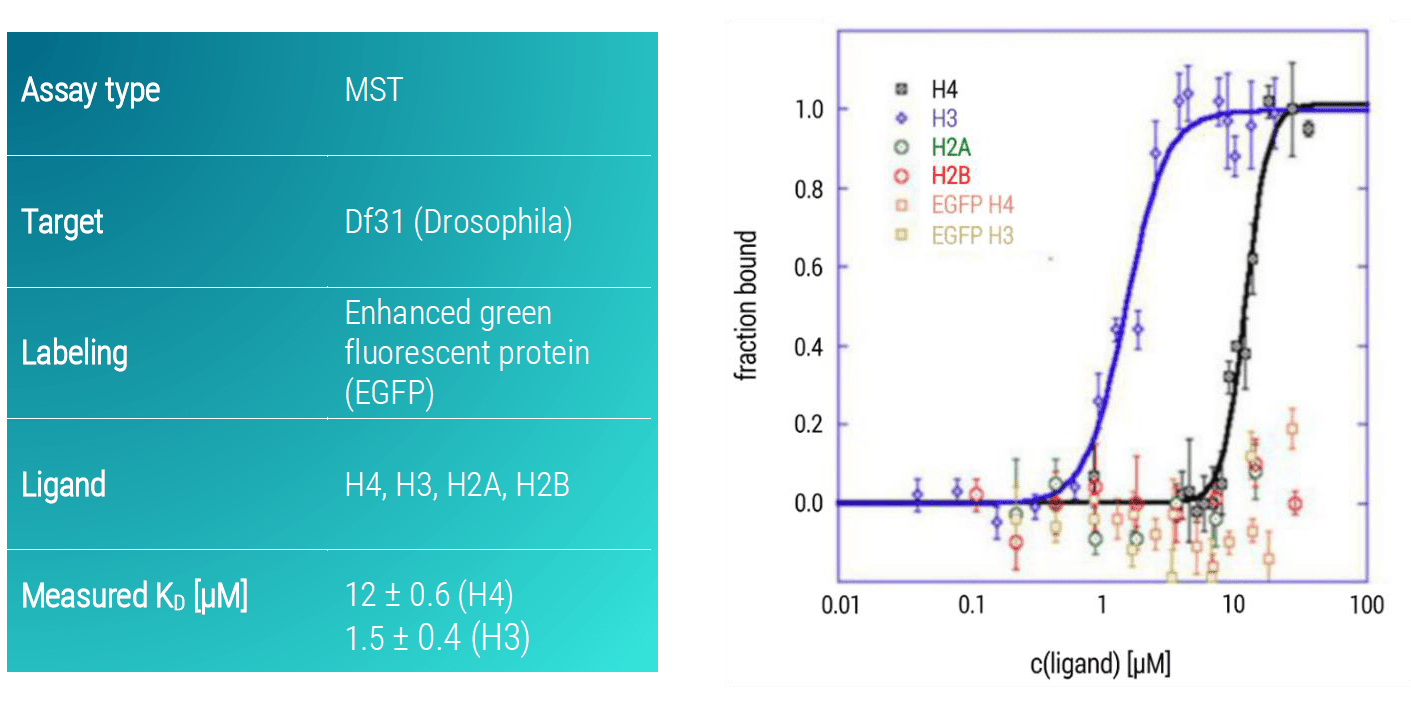
Figure 3. MST dose-response curves of DF31-EGFP and core histone molecules (H4, H3, H2A, H2B). Data represent the mean (+/- standard deviation) from three independent experiments and were fitted to the Hill equation. Free EGFP served as a control for specific binding to H4 or H3.
Example: Protein-protein interactions between Decondensation factor 31 (Df31) and Mononucleosomes
The previous section describes the analysis of interactions between Df31 and core histones. It was shown that Df31 specifically interacts with a selection of core histones. Consequently, the question is whether Df31 also binds to mononucleosomes, which mimic the assembled Drosophila chromatin.
Thus, a similar MST assay was developed in which Df31 was titrated as the ligand against target mononucleosomes from Drosophila (Figure 4). Df31 showed a strong binding affinity towards 601 nucleosomes with a KD of 3.7 μM (± 0.6 μM).
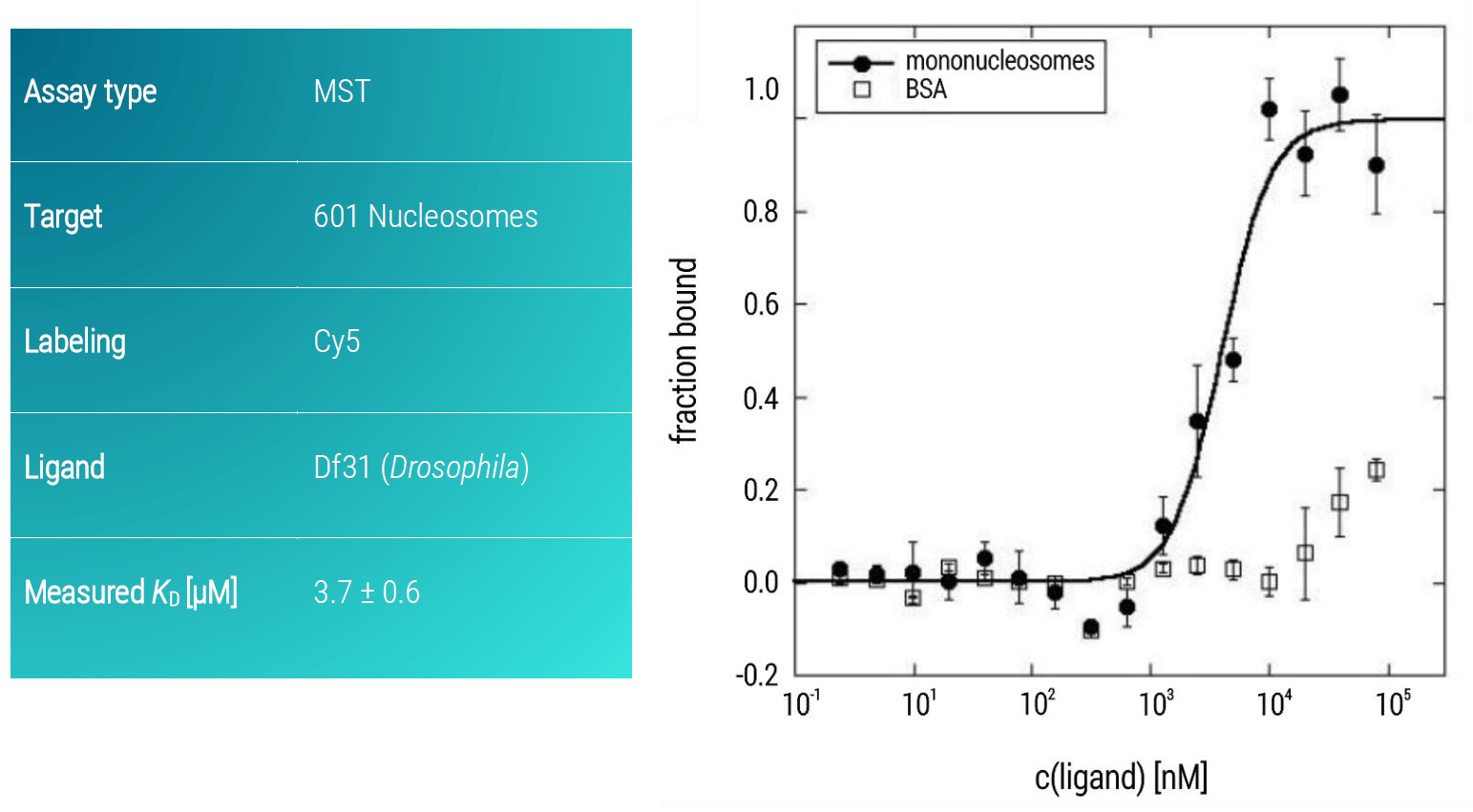
Figure 4. MST dose-response curves of Cy5-labeled 601 nucleosomes and increasing amounts of Df31 protein. Data represent the mean (+/- standard deviation) from two independent experiments and were fitted to the Hill equation. BSA served as control for specific binding.
Protein-Protein Interactions under close-to-native conditions
Example: G-protein coupled receptor vs. monoclonal antibody
G-protein coupled receptors (GPCRs) are a protein superfamily that receive extracellular signals and activate cell-internal signal transduction pathways and responses. Therefore, GCPRs contribute to the development of many diseases and are important targets for drug discovery (more than 30 % of the currently approved small-molecule drugs target members of the GPCR superfamily; Rask-Andersen et al. 2011).
In order to demonstrate the potential of MST for analyzing challenging, trans-membrane GPCRs, we developed an MST assay for studying the interaction of the GPCR CXCR4 (partial) and a monoclonal antibody (mAb). Importantly, the assay was not only designed to work under aqueous buffer conditions but also in 50 % serum, mimicking the a close-to-native bioliquid for a potential GPCR drug. The mAb was used as the target and labeled with an Alexa-647 dye. The GPCR was titrated as the ligand across a concentration range of 1 µM down to 10 pM.
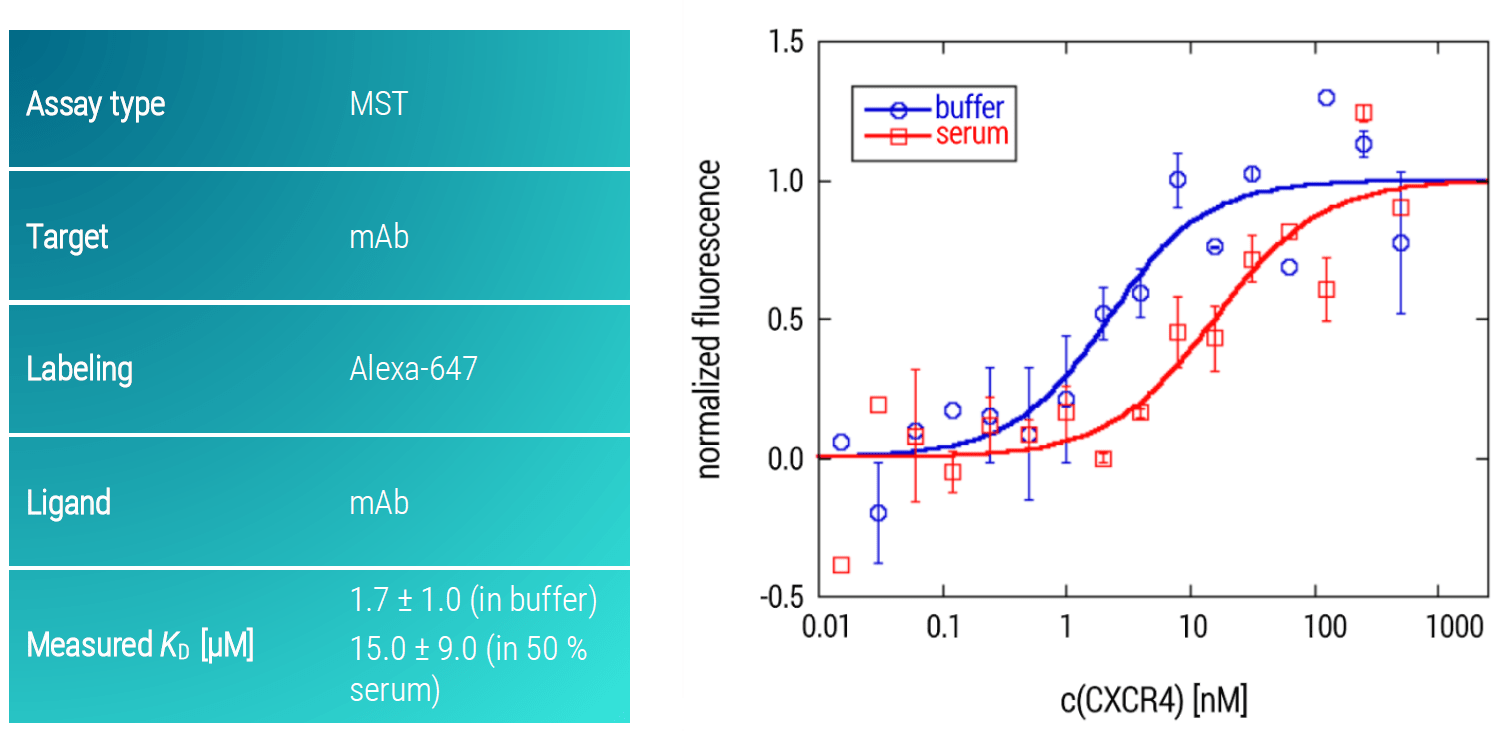
Figure 5: MST dose-response curves of CXCR4 and mAb in buffer and in 50 % serum. Data are fitted to a KD‑binding model under a 1:1 binding situation. Data and error bars are the mean and standard deviation of two independent experiments.
Conclusion: 2bind MST assays for measuring binding of aptamers to proteins
In this application note we show MST assays that allow for the efficient, rapid, and precise characterization of the binding affinities of RNA or DNA aptamer-protein interactions. With only minor tweaks such assays can be adapted for almost all protein-aptamer interactions.
Additionally, this application note highlights the potential of MST assays for discovering previously unseen off-target effects of aptamers in native-like biological solutions.
Click here for a full list of services for aptamer analysis. Alternatively, see our services for protein analysis. If you like to directly contact us for any kind of service, use the contact form below.
Literature
Baaske et al. Optical thermophoresis for quantifying the buffer dependence of aptamer binding. Angew. Chem. Int. Ed Engl. 2010; 49(15): 2238-2241
Breitspeicher et al. Aptamer Binding Studies Using MicroScale Thermophoresis. Methods Mol Biol. 2016;1380: 99-111.
Duhr & Braun. Why molecules move along a temperature gradient. Porc. Natl. Acad. Sci. USA 2006; 103(52): 19678-19682.
Entzian & Schubert. Studying small molecule–aptamer interactions using MicroScaleThermophoresis (MST) Methods. 2016; 97: 27–34
Foley et al. Analysis of conglutin seed storage proteins across lupin species using transcriptomic, protein and comparative genomic approaches. BMC Plant Biology (2015) 15(106): 1-12.
Gupta, Duhr & Baaske. MicroScale Thermophoresis (MST). Encyclopedia of Biophysics. 2018: 1-5
Siller-Matula et al. Thrombin as a multi-functional enzyme. Focus on in vitro and in vivo effects. Thromb Haemost 2011; 106(6):1020–1033
Kohno & Pouyssegur. Targeting the ERK signaling pathway in cancer therapy. Ann Med. 2006; 38(3): 200-211.
Jauset Rubio et al. β-Conglutin dual aptamers binding distinct aptatopes. Anal Bioanal Chem. 2016; 408: 875–884
